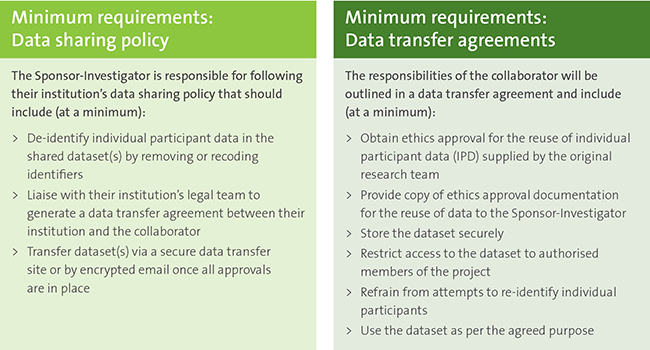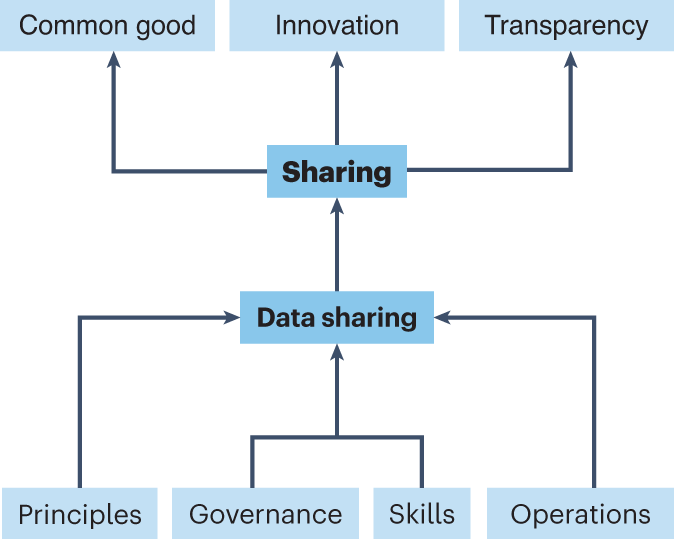
Implementing clinical trial data sharing requires training a new generation of biomedical researchers | Nature Medicine

Sharing and reuse of individual participant data from clinical trials: principles and recommendations | BMJ Open

Individual Patient-Level Data Sharing for Continuous Learning: A Strategy for Trial Data Sharing - National Academy of Medicine
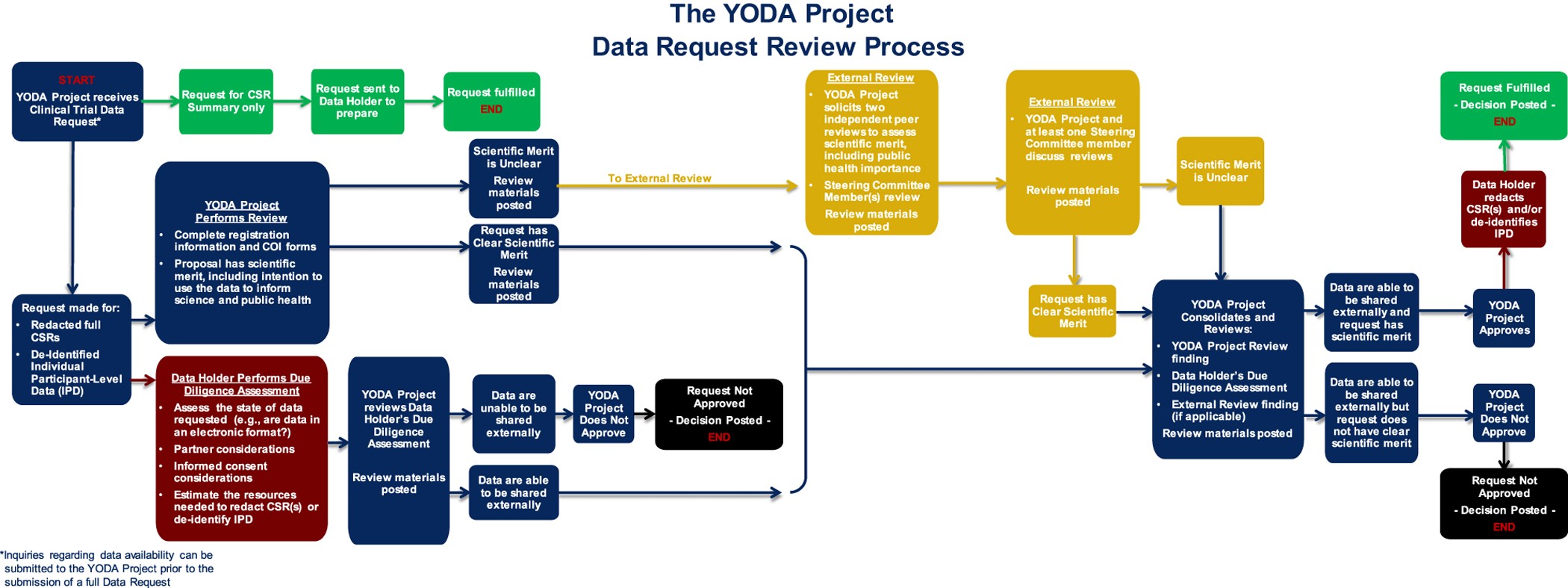
Overview and experience of the YODA Project with clinical trial data sharing after 5 years | Scientific Data
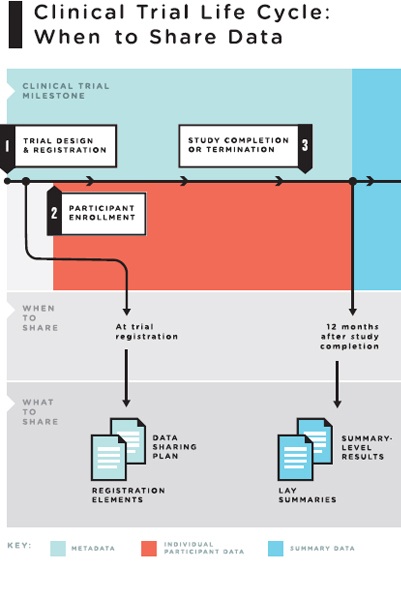
4 The Clinical Trial Life Cycle and When to Share Data | Sharing Clinical Trial Data: Maximizing Benefits, Minimizing Risk | The National Academies Press

Individual Patient-Level Data Sharing for Continuous Learning: A Strategy for Trial Data Sharing - National Academy of Medicine
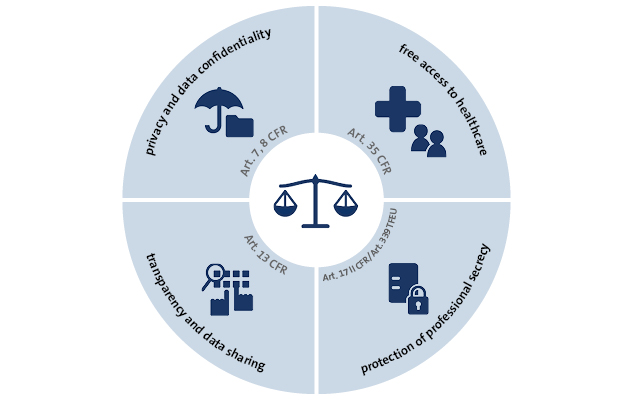
The obligatory sharing of clinical trial data in the European Union - datenschutz notizen | News-Blog der DSN GROUP

Clinical trial data sharing: a cross-sectional study of outcomes associated with two U.S. National Institutes of Health models | Scientific Data